Fuels / Thermochemistry
Enthalpy / 'heat content'
Enthalpy is the ‘heat content that is stored in a chemical system’. It is a term that is can be interchanged with energy when talking about heat changes in a reaction.
We can measure this by measuring the temperature change in the surroundings and carrying out some calculations.
Changes in the chemical system must be equivalent to the surroundings, so we measure the energy changes in the surroundings as a change in temperature.
Heat lost by chemical system = heat gained by surroundings
Heat gained by chemical system = heat lost by surroundings
Δ H (change in enthalpy) = H (products) - H (reactants)
Exothermic reaction – enthalpy of products is less than that of the reactants and so there is a resulting heat loss to the surroundings
Endothermic reaction - enthalpy of products is greater than that of the reactants and so there is a resulting heat being absorbed from the surroundings
Bond breaking is an endothermic process requiring an input of energy and bond making is an exothermic process resulting in a release of energy.
Standard conditions
Enthalpy changes vary according to conditions such as temperature and pressure.
Standard enthalpy changes are given the symbol ΔH and they are given the units kJ mol–1.
*These are the standard conditions:
- Temperature: 298 K (25 °C)
- Pressure: 100 kPa (1 atm)
- Concentration: 1 mol dm–3
*The standard state of a substance is its physical state under conditions of 100 kPa and 298 K.
*Heat of reaction, ΔHr
The heat of reaction is the enthalpy change that accompanies a reaction in the molar quantities expressed in a chemical equation under standard conditions, all reactants and products being in their standard states.
Calorimetry
Calorimetry is used to find the amount of heat released or absorbed during a reaction. A simple method relies on using the heat to warm up a known mass of water by a known amount.
Specific heat capacity (c) is the energy required to raise the temperature of 1 g of a substance by 1 °C.
The equation below is used to work out how much heat is absorbed by the water:
q = m × c × ΔT where q = heat energy, J
m = mass of water, g
c = 4.2 J g–1 K–1
ΔT = change in temperature, K
Note that 100 cm3 of water has a mass of 100 g, c is the specific heat capacity of water, and a change in temperature by 1 °C is also a change by 1 K.
A bomb calorimeter can be used to accurately measure heats of combustion.
Note: if the questions gives the total heat capacity rather than specific head capacity then the mass has all ready been multiplied by the specific heat capacity so you do not need to do this again.
i.e if using 100 g and specific heat capacity is 4.2 J g–1 K–1 then the total heat capacity is 420 J K-1
Energy Profile Diagram
The effect of using a catalyst on rate can be demonstrated using an energy profile diagram. The catalyst provides an alternate route with a lower activation energy.
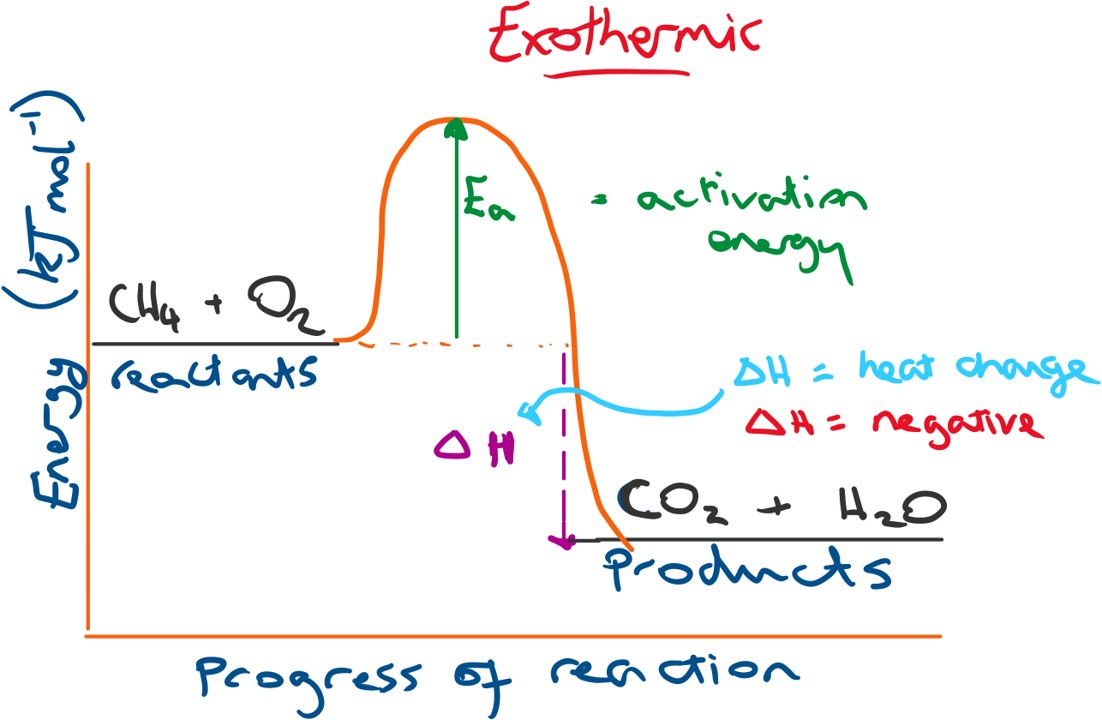
Below is a profile diagram for an exothermic reaction. The change in energy (ΔH) will be negative because the products have a lower energy than the reactants.
N.B. energy change = energy of products - energy of reactants.
This results in that excess energy released into the surroundings after the reaction and so we observe a heat or energy gain for the surroundings
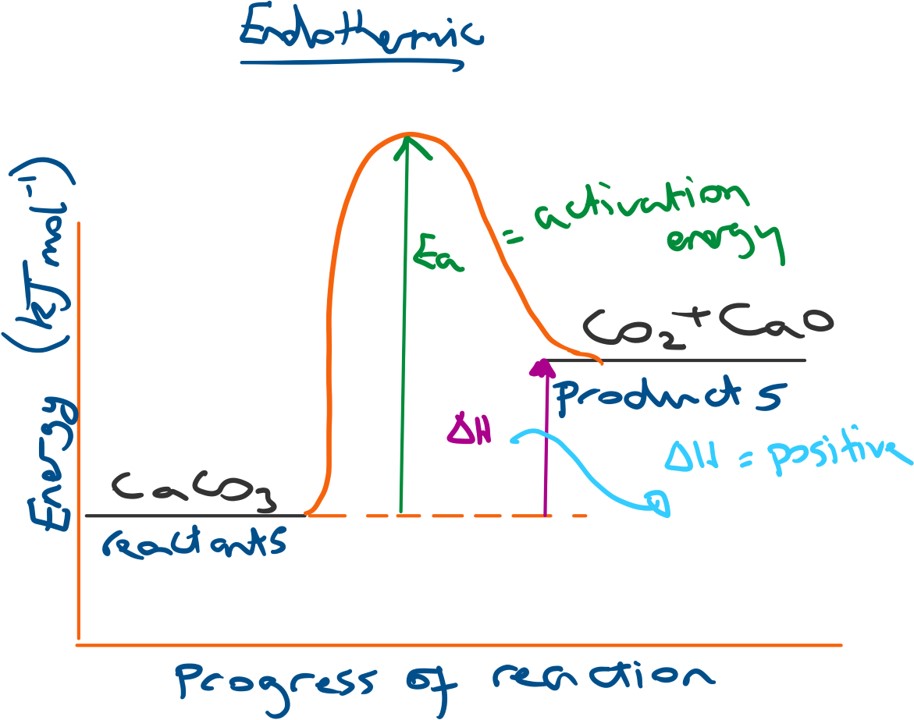
For an endothermic energy profile diagram the products will have a higher energy than the reactants and so the overall change in energy (ΔH) will be positive (thus energy is taken into the system from the surroundings, resulting in heat loss).
This results in energy being brought in from the surroundings and so we observe a heat or energy loss from the surroundings.
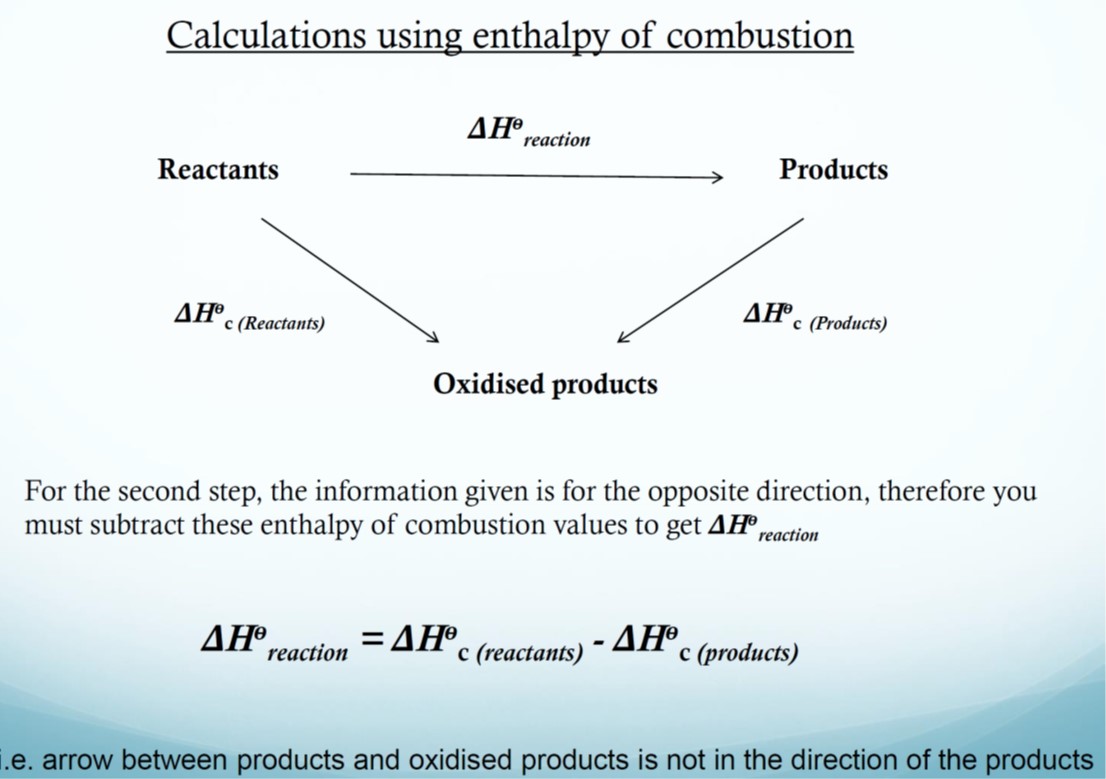
Hess Cycles
Hess’ law: If a reaction can take place by more than 1 route and the initial and final conditions are the same, the total enthalpy change is the same for each route.
A Hess cycle is a diagram showing alternative routes between reactants and products which allows the indirect determination of an enthalpy change from other known enthalpy changes using Hess’ law.
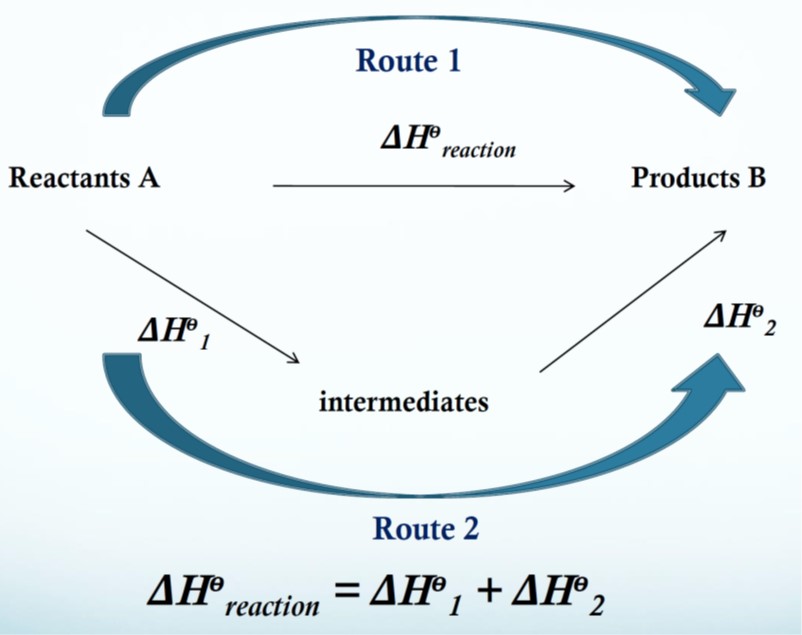
If you know two of the energy changes for the above reactions then you can calculate the third.
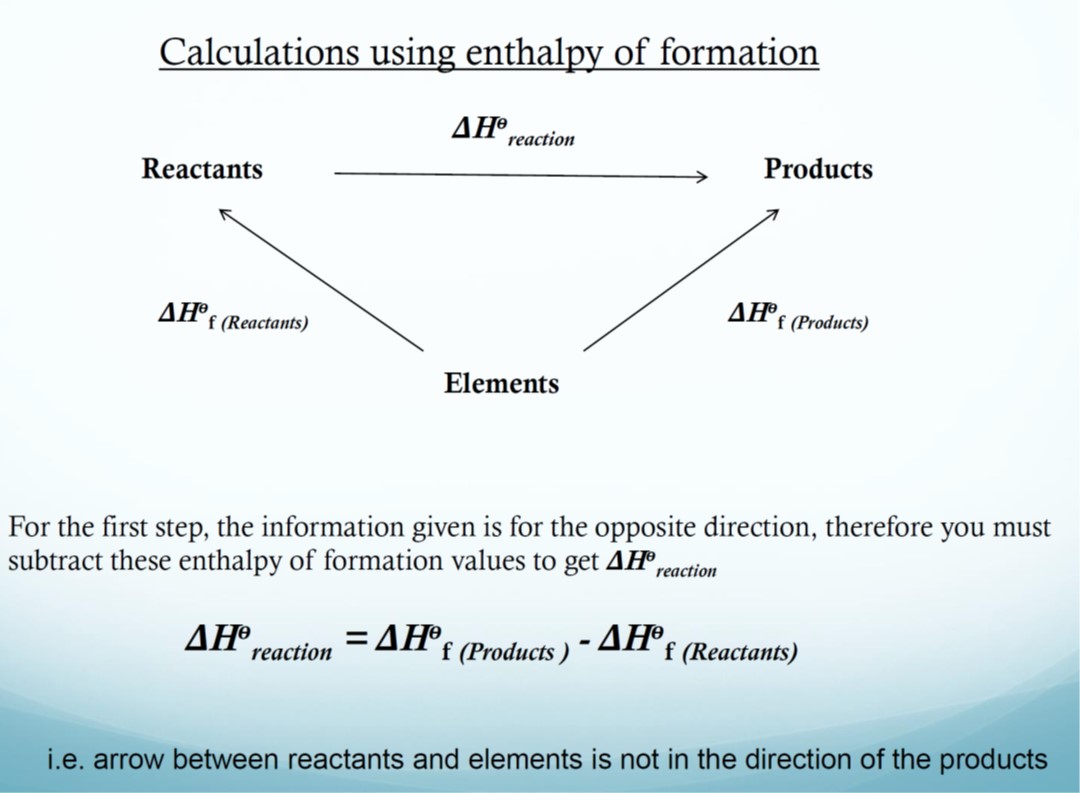
*Heat of formation, ΔHf
The heat of formation is the energy change that takes place when 1 mole of a compound is formed from its constituent elements in their standard states under standard conditions.
The heat of formation of an element in its standard state is zero.
Heat of combustion, ΔHc
The heat of combustion is the energy change that takes place when 1 mole of a substance reacts completely with oxygen under standard conditions, all reactants and products being in their standard states.
© Copyright rmcstudy