Rates introduction -
Collision theory
For a chemical reaction to take place the reacting particles have to collide with enough energy. The energy that is required is called the activation energy. If they do not collide with enough energy then the reaction will not take place.
Activation Energy : The minimum energy required to start a chemical reaction by the breaking of bonds.
Collisions must not only occur with sufficient energy (activation energy), they must occur with in an appropriate orientation. (Head on collisions will be more successful than glancing collisions, usually referred to as a steric effect).
Any factor that will increase the frequency of collisions will increase the rate of reaction.
1) Temperature
Temperature is proportional to the kinetic energy of the particles. The more energy that the particles have then the faster they move around.
An increase in temperature results in the particles having higher kinetic energy leading to more frequent collisions and also more successful collisions as the collisions themselves occur with more energy.
At a rough approximation a 10 C increase in temperature doubles the rate of reaction.
2) Concentration
This is the amount (number of moles) of a given substance in a certain volume of solvent.
An increase in concentration results in more particles per unit volume, hence more frequent collisions between the particles.
3) Surface area
This tells you about the relative amount of solid particles that are exposed for reaction with other particles.
Surface area – More exposed particles leads to more frequent collisions and so more successful collisions.
An example of this is when powdered solids are used in place of lumps of solid. (e.g. powdered CaCO3 vs marble chips)
4) Catalyst
This is a substance that increases the rate of reaction without being consumed in the process.
It works by providing an alternative route with a lower activation energy.
Homogeneous catalysts are in the same phase as the rest of the reactants. Enzymes are an example of biological catalysts.
Another example is when Cobalt (II) chloride (which forms the PINK [Co(H2O)6]2+ complex in water) catalyses the decomposition of tartrate ions by hydrogen peroxide (forming a green intermediate - Co3+). The pink colour returning indicates the end of the reaction.
Heterogenous catalysts are in a different phase to the reactants. They usually work via the surface adsorption theory, where gas particles usually are adsorbed on the the surface of the catalyst. There, the bonds in the reacting particles are weakened by interactions with the catalyst, helping to speed up the reaction. The products then leave viaI desorption.
5) Nature of reactants
Ionic substances are dissociated in water and so will react very quickly as ionic bonds need not be broken.
If a covalent bond is required to be broken then the reaction will be slower.
Calculating rate
rate of reaction = change in concentration of reactant or product divided by the time it takes for the change to take place.![]()
As reactions proceed fewer collisions take place per second and so the reaction slows down.
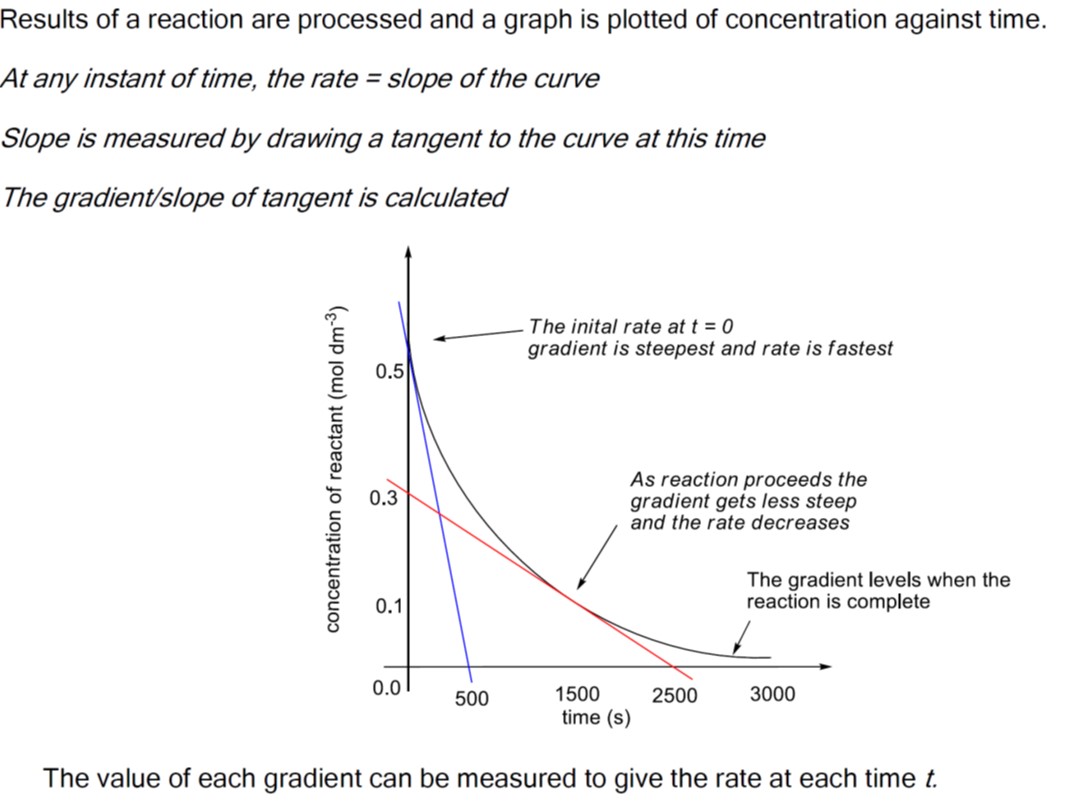
Remember that the rate of change can be calculated using the formula for the slope of a line in coordinate geometry, i.e. the difference in the y-values divided by the difference in the x-values.
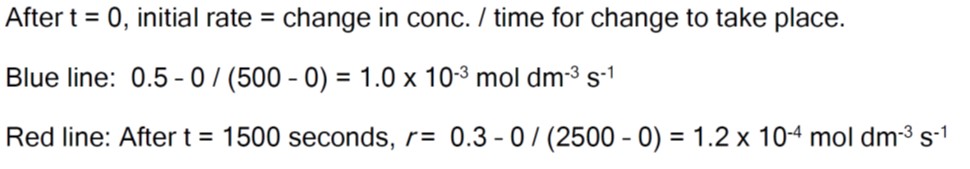
*hence rate is higher (reaction faster) at the beginning.
Energy Profile Diagram
The effect of using a catalyst on rate can be demonstrated using an energy profile diagram. The catalyst provides an alternate route with a lower activation energy.
Below is a profile diagram for an exothermic reaction. The change in energy will be negative (thus released into the surroundings resulting in heat gain) because the products have a lower energy than the reactants.

An endothermic energy profile diagram will be the same as above but the products will have a higher energy than the reactants and so the overall change in energy will be positive (thus energy is taken in from the surroundings resulting in heat loss).
© Copyright rmcstudy