Basic atomic theory
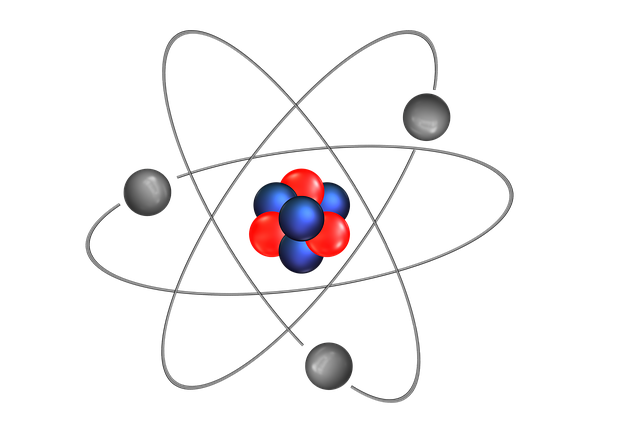
All substances are made from atoms, and any given element always has atoms with the same number of protons and electrons.
Atoms have a small central nucleus (containing protons and neutrons) which is 'orbited' by electrons. Some properties of each can be found here:

The atomic number (smaller number in the periodic table) shows the number of protons (and therefore, the number of electrons as all atoms have no overall charge) in an atom.
The mass number (larger number in the periodic table) shows the number of protons and the number of neutrons in an atom.
Atomic Emission Spectra
Atomic emission spectra provide evidence that electrons are arranged in energy levels. An energy level is the discrete (fixed) amount of energy that an electron possesses. A continuous spectrum of colours are seen when white light passes through a prism. We can see the actual colours because their corresponding frequencies are found in the visible section of the electromagnetic spectrum.
When energy is supplied to individual elements they emit a spectrum containing emissions of a particular wavelength. This is called a line spectrum. The following process occurs:
- Electrons gain energy and move from their lowest available energy level (ground state) to a higher available energy level (excited state).
- Electrons themselves occupy a fixed energy level, and so have a fixed quantum of energy.
- As the electron drops back down to lower available energy level, a fixed amount of energy is emitted as a photon of light, which shows as a line in the emission spectrum.
- When the electrons drop to the second energy level, that frequency of light is emitted in the visible section. This is called the Balmer series.
E2- E1= hf
where E2-E1 is the difference in energy between the excited state and energy level n =2
h = Plank’s constant
f = frequency of light
Ionisation energy
Evidence for the model of an atom can be found from ionisation energy. This is the energy required to remove one mole of electrons from one mole of atoms in their gaseous ground state (kJ mol-1). (Essentially measures how hard it is to remove an electron from an atom/ion).
Atoms and ions will have different first ionisation energies because each will have a different nuclear attraction to their outershell electrons. They do so because of atomic radius, nuclear charge and electron shielding.
Atomic radius (a.r.) is half the distance between the nuclei of two of the same atoms joined together by a single covalent bond. As a.r. gets smaller then nuclear attraction increases.
Nuclear charge (n.c.) is essentially the number of protons in the nucleus. As n.c. gets larger then nuclear attraction increases.
Electron shielding (e.s.) is the repulsion between inner shells of electrons and the outershell.The fewer the number of shells then the larger the nuclear attraction.
A higher a.r. and e.s. will offset a higher n.c.
Atomic orbital introduction
Ionisation energies provide evidence that energy levels are split up into sub-levels which gives rise to the theory of atomic orbitals.
Atomic orbital shape
Atomic orbitals are regions of space where an electron is most likely to be found. Analysis of the position of an electron at any one moment in time gives rise to a model which can account for the shape of this region.
The atomic orbitals necessary for leaving cert chemistry are s and p orbitals. There are one s and three p orbitals (which differ only in their orientation) in every main energy level (bar the first, which only has two electrons). The number of electrons on each main energy level can be found by calculating two 'n' squared where n is the number of the energy level ( = principal quantum number).
© Copyright rmcstudy