Chemical Bonding
Ionic Bonding & structure
Covalent bonding
electronegativity & intermolecular forces
Shapes of molecules
Oxidation and reduction
Ionic bonding
Theionic bond is the electrostatic interaction between oppositely charged ions.
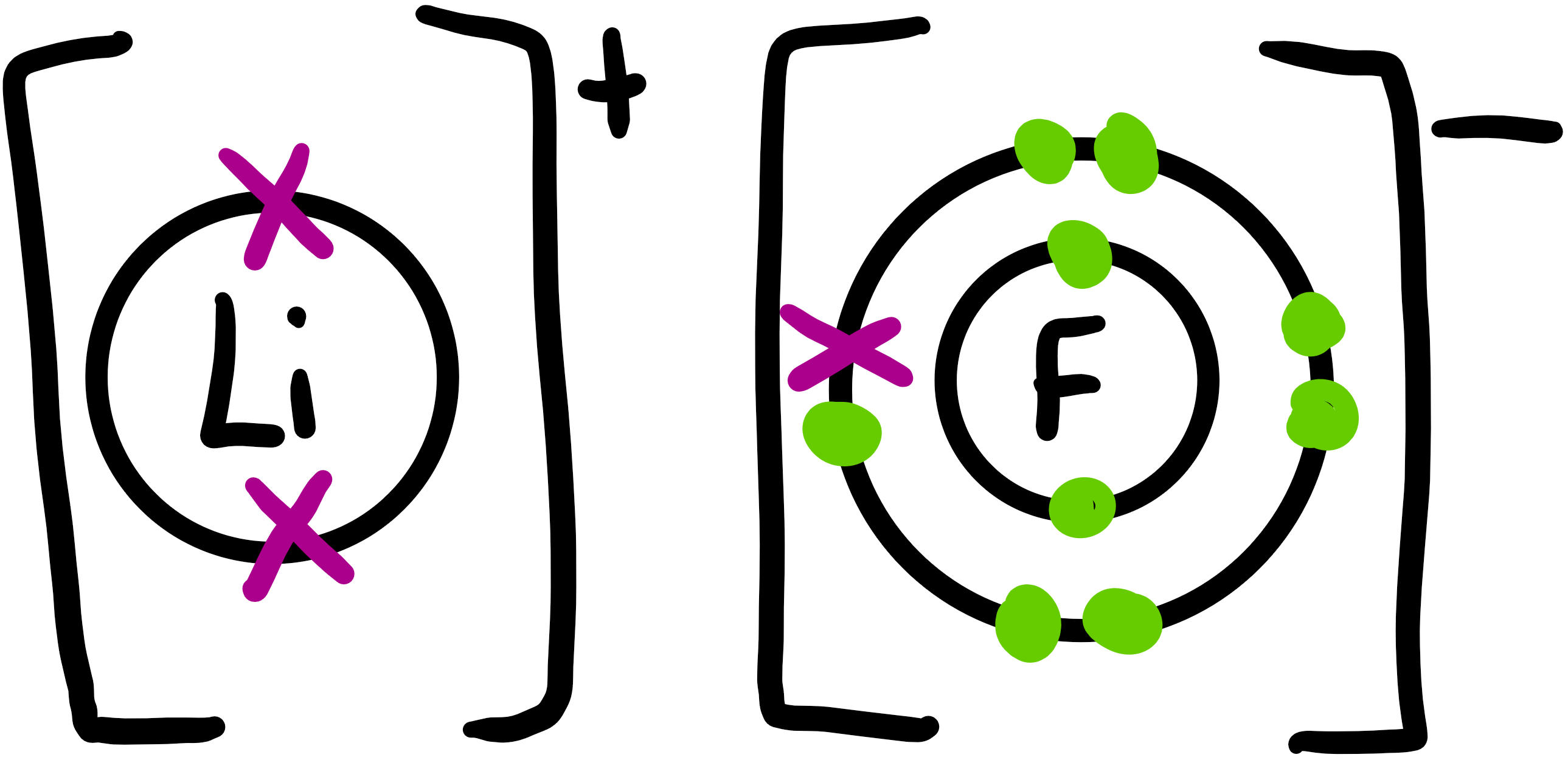
It involves the transfer of electrons from a metal atom to a non-metal atom (usually - electronegativity difference greater than 1.7 between the two atoms leads to the ionic behaviour of a bond).
When an atom loses/gains an electron it becomes an ion as it has an electrostatic charge on it due to uneven number of protons/electrons.
Metals usually lose electrons to become positive ions.
Non-metals usually gain electrons to become negative ions.
Ionic structure
Ionic compounds consist of a huge lattice of positive and negative ions packed in a regular pattern. There are very strong electrostatic attractions between the ions (ionic bonds). This structure give rise to a number of physical properties of ionic compounds.
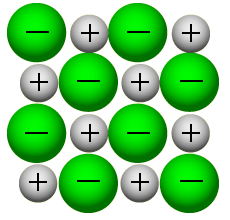
- high melting and boiling points - strong electrostatic forces between oppositely charged ions.
- crystalline - regular array of ions in the lattice.
- brittle – any distortion brings ions of the same charge adjacent to each other => they repel.
- most are soluble in water (except some e.g. MgO).
- conduct electricity when molten or in solution.
Covalent bonding
Covalent bonding involves the sharing of a pair of electrons between two non-metal atoms.
The bonds are strong and are found in molecules, e.g. hydrogen and water, or giant covalent structures like diamond and graphite.
Covalent bonding is a strong attraction between the bonding pair of electrons and the nuclei of the atoms involved in the bond.
A covalent bond is a shared pair of electrons.
The octet rule is usually obeyed when atoms form bonds. This states that atoms have a tendency to adopt a noble gas configuration when bonding due to stability of configuration. Exceptions include boron and silicon compounds.
A dative covalent bond occurs when one of the two atoms supplies both electrons in the shared pair.
Not discussed in video:
A sigma bond is a bond formed by the head-on overlap of two orbitals. A pi bond is a bond formed by the sideways overlap of two p orbitals.
Electronegativity
Electronegativity is a measure of an atoms ability to attract the shared pair of electrons in a covalent bond. Atoms that are bonded which have an electronegativity difference greater than 0.4 results in a polar covalent bond.
Intermolecular forces
Intermolecular forces are forces of attraction between compounds that are molecules. They are responsible for the way most substances interact with each other, particularly in living organisms.
There are three types:
1) Permanent dipole-dipole interactions are those in which a permanent dipole in a molecule is attracted to another molecule in which there is a permanent dipole of the opposite charge.
2) Van der Waals forces (VdW)are forces of attraction between molecules which are also known as induced dipole-dipole interactions. A non-polar bond exists when the electronegativity difference between the two atoms in the bond is less than 0.4. The electron density between the atoms fluctuates causing an instantaneous temporary dipole in the molecule, which then induces a dipole in a neighbouring molecule. The opposite dipoles attract which is the Van er Waals force. As the number of electrons increase so do strength in VdW.
3) Hydrogen bonding occurs when there is a strong permanent dipole-dipole interaction between an electron-deficient hydrogen atom on one molecule and a lone pair of electrons on a highly electronegative atom (only Nitrogen, Oxygen or Fluorine) in a different molecule.
Shapes of Molecules
The shape of all molecules / ions are dictated by the number and type of electron pairs present in the molecule according to the Valence Shell Electron Pair Repulsion theory (VSEPR).
The distribution of electron pairs around a central atom will dictate the shape of simple molecules. Electron pairs can either be bonding pairs ((b.p.), i.e. a typical covalent bond) or lone pairs (l.p.) of electrons. Lone pairs of electrons occupy orbitals that sit closer to the central atom than bonding pairs (which occupy molecular orbitals shared between two atoms); this means that they take up more space around the central atom.
To work out shape of molecule, just note the number and type of electron pairs around the central atom. The shape of molecule will be one of the following:
Number and type of electron pair - shape - bond angle
1 b.p. - linear - 180
2 b.p. - linear - 180
3 b.p. - trigonal planar - 120
2 l.p. & 2 b.p. - v-shaped - 104.5
3 b.p. 1 l.p. - pyramidal - 107
4 b.p. - tetrahedral - 109.5
5 b.p. - trigonal bipyramidal - 90 & 120 (not on lc course)
6 b.p. - octahedral - 90 (not on lc course)
Oxidation and Reduction
Oxidation can be considered in a number of ways. In a reaction with oxygen, a substance that gains oxygen has been oxidised and a substance that loses oxygen is reduced.
In electron transfer reactions, oxidation is the loss of electrons by a species and reduction is the gain of electrons (OIL RIG).
Another method for identifying oxidation is to assign oxidation numbers to species in a reaction. Oxidation occurs in this context when an element has increased its oxidation number on going from reactant to product. Conversely, reduction occurs when an element has decreased in oxidation number.
The oxidation number of an atom is a measure of the number electrons lost or gained by an atom in a compound compared to the uncombined atom. These numbers are assigned using the following rules:
Species Oxidation number example
- Uncombined element 0 C, Na, hydrogen, fluorine
- Simple ion charge on ion Na+ =+1, Cl- = -1
- Compound total oxidation number = 0 methane, water
- Polyatomic ion total oxidation number = charge on ion sulfate = -2
- Combined oxygen -2 H2O, CaO
- Combined hydrogen +1 H2O, CH4
- Combined fluorine -1 NaF, AlF3
Exceptions: Oxygen becomes +1 when bonded to fluorine. -1 in peroxides. When bonded to a metal H becomes -1 (hydride ion)
Not discussed in the video: Disproportionation occurs when an atom simultaneously undergoes both oxidation and reduction in a reaction. E.g. when chlorine (O.N = 0) is added to water, Hydrochloric acid (HCl, O.N. = -1) and chloric (I) acid (HOCl, O.N. = +1) are formed.
Using half equations to balance REDOX equations
© Copyright rmcstudy