Organic Chemistry
Introduction & key definitions
Nomenclature &
Homologous series
Crude oil &
Hydrocarbons
Halogenoalkanes
Alcohols
Carbonyls
Mechanisms for LC
Introduction & key definitions
Organic chemistry is the chemistry of carbon based molecules. Most molecules we deal with in life (such as medicinal drugs & DNA for example) are built from carbon, and also contain hydrogen, and quite often oxygen and nitrogen; a few examples of common compounds can be seen below (they are represented using skeletal formulae - see intro video):

A few definitions are important to begin with due to the huge numbers of organic compounds:
A Homologous series is a family of compounds with the same functional group (part of molecule responsible for chemical reactions) and the same general formula.
Some molecules will have one functional group, while many will have multiple, and these groups can interact with various different molecules using the forces of interaction discussed in the covalent structure section.
In a homologous series, each successive member will differ by one carbon atom and two hydrogen atoms, so the extra CH2 group causes that member to have stronger Van der Waals forces and so a higher boiling point than its preceding member.
Representing molecules
Molecular formula (MF) shows the actual number of atoms of each element in a molecule.
Empirical formula (EF) shows the simplest whole number ratio of atoms of each element present in a compound.
Structural formula shows the relative positions of atoms in a molecule e.g. for butane: CH3CH2CH2CH3 or CH3(CH2)2CH3,
Displayed structural formula shows the relative positioning of all the atoms in a molecule and the bonds between them. You need to be very familiar with this for LC chemistry.

Skeletal formula is a simplified organic formula with carbons and hydrogens not shown, leaving the main chain with associated functional groups. Hydrogens are shown attached to atoms other than carbon (see first video for detailed explanation).

Structural isomers are molecules that have the same molecular formula (same number and type of atoms) but different structural formula (different arrangement of atoms). For example, the following are structural isomers of a molecule called pentane.
Nomenclature
This is a naming system for organic compounds created by the International Union of Pure and Applied Chemistry (IUPAC). The name can consist of three parts, although not all three are always necessary.

The root of the name depends on the number of carbons in the longest unbranched chain of carbon atoms. e.g. If a molecule has one carbon and it is from the alkane family then it must be methane (see homologous series below).

Sometimes 'an' is placed in between the root and the suffix. e.g. methanol.
Homologous Series
Below is a list of all the homologous series that are required for the leaving cert (except knowledge of benzene).





Crude oil
Crude oil is a mixture of hydrocarbon molecules (carbon and hydrogen atoms only -discussed in the next section).
The soft tissue of plants and animals which had died millions of years ago was changed under high pressures and temperatures (from layers of sediment) to form crude oil.
It is not very useful in its crude state so it gets purified using fractional distillation which is the separation of molecules into different fractions based on their boiling points. Longer chain hydrocarbons will have higher boiling points due to the increased Van der Waals forces holding the molecules close together.
- Oil is preheated.
- Hot at the bottom and cold at the top.
- Vapours rise up the column and then condense at different heights depending on their boiling point
You need to know the fractions and their uses:

Hydrocarbons - Introduction
Hydrocarbons - Chemical reactions
Hydrocarbons - notes
Hydrocarbons are molecules that contain hydrogen and carbon atoms only. There are 3 types that you need to know for L.C. chemistry: Alkanes (all single bonds), Alkenes (at least one C=C double bond) & Alkynes (CC triple bond).
Alkanes
These are saturated (contain only single bonds) hydrocarbons and are the simplest homologous series. Importantly they make up the backbone of most organic molecules (as most molecules will have at least one other functional group).

All alkane molecules follow the general formula. It can be seen that each successive member has an extra CH2 group. This has implications for the size of the molecule and for physical properties like boiling point. A bigger molecule means more points of contact and so stronger overall Van der Waals forces of attraction between the molecules, leading to a higher boiling point as the chain length increases.

Chemical reactions
Solubility: As we know, oil and water are not miscible. The reason being that oil based molecules (non-polar) interact with eachother using Van der Waals attractions and water based molecules (polar) interact using hydrogen bonds.
Combustion - rapid reaction of a substance with oxygen releasing energy. Complete combustion of any organic molecule will always produce carbon dioxide and water. Incomplete combustion (insufficient oxygen) produces carbon monoxide (and soot) and water. Carbon monoxide is toxic as it irreversibly binds to the haemoglobin in the blood.
Shorter alkanes burn more cleanly and efficiently than longer chain molecules.
To balance combustion reactions always begin with balancing carbon, then hydrogen, then oxygen atoms.

Alkanes also undergo free radical substitution - see mechanisms for L.C. section.
Alkenes
Unsaturated molecules: Carbon-carbon double bond present.

Most organic molecules can trace their roots from crude oil, and ethene is no different. Catalytic cracking of longer chain molecules isolated from fractional distillation of crude oil using an Aluminium oxide catalyst can produce ethene.
It can also be made synthetically from the dehydration of ethanol using aluminium oxide as a dehydrating agent. This is a popular question on L.C. chemistry. Glass wool is also used to absorb ethanol. Once heat is applied ethanol vapour gets passed over the catalyst and dehydrated. This is an example of an elimination reaction.


N.B.: Test for unsaturation: bromine water (orange colour) is added and this turns colourless when an alkene is present.

The two covalent bonds in the double bond are formed from a different orientation of orbital overlap. One of the bonds (sigma bond) arises from a head-on overlap of orbitals which results in a very strong covalent bond. The other bond (pi bond) however arises from a side-on overlap of orbitals (hybridisation is not covered in L.C. chemistry). This weaker bond results in the chemistry that they undergo, i.e. Addition reactions.

Addition polymerisation: Double bond breaks and a new carbon-carbon single bond forms. Under certain conditions, alkenes (monomers) can undergo polymerisation reactions with themselves to form very long chain molecules known as polymers.

See mechanisms section for more on chemical reactions of alkenes.
While not on the course, the last point to mention about alkenes is the fact that you can get geometrical stereoisomers. Stereoisomers are molecules whose atoms are connected in the same way but have a different arrangement in space. Geometrical ones have groups attached to different sides of the double bond. e.g.:

Alkynes
Alkynes are hydrocarbons that have a carbon-carbon triple bond. The only one on the L.C. course is the first member, ethyne, also known as acetylene ( used in blow torches for welding). The formation of ethyne from water being dropped onto calcium carbide is a popular question.
![]()

Halogenoalkanes
These are compounds which have a halogen bonded to a carbon atom somewhere in a molecule. While they are really useful compounds (particularly as organic solvents - they can help dissolve a large variety of compounds) they are not examined to a great extent on the L.C. course. Naming is important as is the formation of them using the two methods described in the mechanism section. A very useful reaction known as nucleophilic substitution is not covered on the L.C. course.

Alcohols
A hugely important class of compounds, it is important to know how to make the most common alcohol, ethanol, and understand some of its reactions. There are two common methods of formation. The quickest and most efficient is the hydration of ethene, where water is added to ethene using an acid catalyst (phosphoric acid usually).

The other method and more common from an alcohol production point of view is the anaerobic respiration (fermentation) of glucose, producing carbon dioxide and ethanol. The yield of ethanol is low but this brewing method is more sustainable as glucose comes from plant material, whereas ethene is usually generated from crude oil.

The alcohol functional group is represented by ![]() where R is a generic group beginning with a carbon atom, usually representing an alkyl (alkane) chain:
where R is a generic group beginning with a carbon atom, usually representing an alkyl (alkane) chain: ![]() . The naming system for the first two members of the homologous series is fairly standard:
. The naming system for the first two members of the homologous series is fairly standard:

but we need to classify alcohols into different types when we get to 3 carbons:

A third classification comes in when we look at an isomer of butanol:

Physical properties
Boiling points, which are directly related to the strength of the intermolecular forces, are relatively high for low molecular weight compounds, due to the strong hydrogen bonds.

A Hydrogen bond (H-bond above) is an electrostatic attraction between an electron deficient hydrogen atom (one bonded to a suitably electronegative element, only nitrogen, oxygen or fluorine) and the lone pair on another atom, usually nitrogen, oxygen or flourine. This intermolecular attraction is far stronger than Van der Waals forces and is the reason that alcohols have reasonably high boiling point. As the chain length of the alcohol increases then the boiling point will also slightly increase as there are more Van der Waals forces of attraction.
Solubility:
Small chain alcohols are very soluble in water as both interact well via hydrogen bonds (caol le caol agus leathan le leathan; like dissolves like). As the chain length increases then they become less soluble as the non-polar alkyl chains don't interact well with the polar water molecules.
Reactions of Alcohols
The main reactions that are necessary to know for L.C. chemistry is the reaction with an acid to form an ester in a substitution reaction (see ester section below), the dehydration of an alcohol to from an alkene in an elimination reaction (see alkene formation) and the oxidation of alcohols to form aldehydes, ketones or carboxylic acids.

Oxidation will happen if there is a hydrogen atom attached to the carbon bearing the OH group.

As the oxidising agent above is a dichromate ion, it has an orange colour, as it oxidises the alcohol, it itself gets reduced to chromium ions which are green. So the colour change for the reaction is orange-> green (note the counter ion 'Na+' in 'Na2Cr2O7' above is irrelevant, it could also be 'K+').

The alcohol functional group is mildly acidic as the molecule can be deprotonated (see acids section) to form a salt and hydrogen gas when treated with a reactive metal in a similar manner to carboxylic acids but they are not as acidic.
Carbonyls
There are 4 types of carbonyl that are examined in L.C. chemistry; aldehydes, ketones, carboxylic acids & esters.

Aldehydes
The R-CHO functional group (as opposed to R-COH which represents alcohols) is what makes something an aldehyde. As there is a carbonyl bonded to a hydrogen atom, they must occur at the end of a carbon chain.

As they do not have a suitably electron deficient hydrogen atom, they do not form hydrogen bonds between their molecules. As a result they have relatively low boiling points.

The lone pair on the oxygen atom allows them to form hydrogen bonds to water molecules and so small chain aldehydes are soluble in water. As with ketones, they can be reduced to alcohols using sodium borohydride.
Ketones
Ketones are much like aldehydes but the carbonyl group is 'internal' and not on the end of a carbon chain. Hence the smallest ketone is propanone (3 carbon atoms), that and butanone are the only ones on the L.C. course. Their physical properties and chemical reactions are similar to aldehydes. They can be reduced to alcohols using Hydrogen and a Nickel catalyst, which is also used to make alkanes from alkenes (this reduction of carbonyls is more popular on the mark schemes than the usual sodium borohydride or lithium aluminium hydride).

Carboxylic acids

The generic formula for this class of compounds is R-COOH. All standard acids (not counting lewis acids) provide a source of H+ ions in solution and these are no different. They are weak acids which mean that they don't fully dissociate when in water, only partially dissociate into its ions (see equilibrium):

Carboxylic acids have high melting and boiling points and are soluble in polar solvents like water. They can form hydrogen bonds between like molecules and also with water.
Reactions
As they are acids they will react in the same way as standard acids, although not with the same intensity as they are weak acids. e.g. Ethanoic acid reacts with sodium hydroxide to form its salt (sodium ethanoate) and water. They can react with metals to form salt and hydrogen and react with metal carbonates to form a salt, carbon dioxide and water.

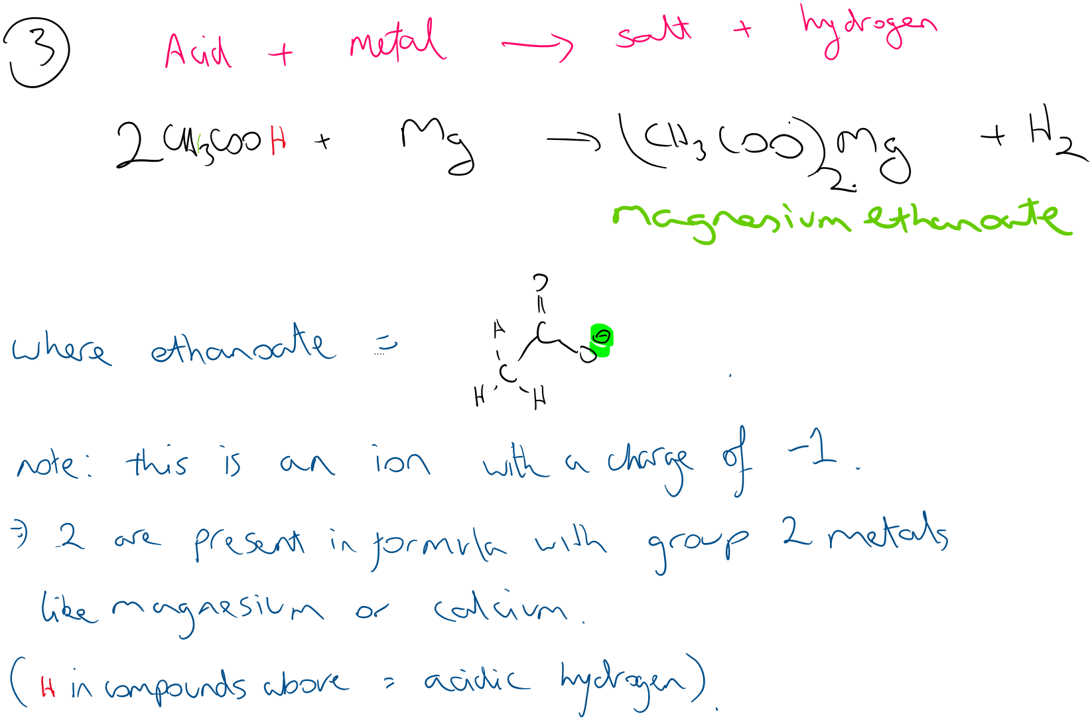
Under acidic conditions (strong acid) they react with alcohols to form esters.
Esters

They have a pleasant smell and are used in perfumes. They have a low boiling point - they cannot form H-bonds with each other (no suitably electron deficient hydrogen atom) but they can with water and so small chain esters are relatively soluble in water.
There are two parts to their name, the first always comes from the alkyl part of the alcohol and the second comes from the carboxylic acid used to make them.
The above reaction is actually reversible. The reverse reaction is used to make biodiesel.
Mechanisms for Leaving Certificate Chemistry
Free radical substitution
Electrophilic / ionic addition
Free radical substitution: This is one of the two mechanisms you are required to know for L.C. chemistry (the other is electrophilic addition to an alkene). A mechanism is a sequence of steps that show the movement of electrons between atoms.
To make new bonds, usually, older covalent bonds need to be broken. They can either break by one of the atoms accepting both of the electrons (the more elctronegative atom will take the electrons) which is known as heterolytic fission.

Homolytic fission is where there is an even breaking of the covalent bond in which both atoms regain one electron to form a species known as a free radical.

Free radical substitution
The following is the mechanism for the free radical substitution reaction of methane with chlorine. (Note: only the purple depictions below are necessary for LC chemistry, the orange writing is an aid to understand what actually happens).

Initiation
It starts with an initiation process where by ultra-violet (UV) light causes a chlorine molecule to split apart by homolytic fission.

Propagation 1
The chlorine radical then extracts a hydrogen atom from a methane molecule, forming a new H-Cl bond and breaking a H-C bond to from a methyl radical.

Propagation 2
The methyl radical then reacts with an chlorine molecule to reform the chlorine radical (chain process as the radical is reformed) and the chloromethane product.

Termination
There are three potential termination steps; essentially a radical must react with another radical so that no new radical is formed. 2 things are worth noting here.
1) The presence for ethane is evidence for this particular mechanism as it has to be formed from two methyl radicals combing
2) The presence of tetra-methyl lead (not shown) or tetra-ethyl lead speed up the reaction by providing extra methy/ethyl radicals.
Electrophilic addition
The next mechanism involves heterolytic fission of a covalent bond in an electrophilic addition mechansim.

At first, the rich electron density on the alkene double bond, induces a dipole in the bromine molecule (see below). The alkene then reacts with the partially positive bromine causing the Br-Br bond to break. The pi bond in the alkene is weaker than the sigma bond (side on overlap of orbitals rather than head on overlap).
There is debate as to the best way to represent the mechanistic approach to form the brominium ion intermediate (the orange dotted line from the bromine to the carbon need not be shown). The bromide ion then attacks the carbon bearing the brominium ion forming a new covalent bond. (terms - nucleophile and electrophile are not specifically on the course).
4 key steps:
1) polarisation of bromine. electron rich nucleophile (nucleus loving) alkene attacks bromine.
2) heterolytic fission of bromine
3) brominium / carbonium ion formation
4) ionic addition of bromide

You will get marks for drawing the carbonium ion (acceptable method) which then gets attacked by the electron rich bromide ion.

In fact, this may be easier to remember as it is also used in the addition of Hydrogen bromide to an alkene.

N.B.: if the alkene is unsymmetrical then the carbocation (C+) forms on ion the carbon atom that has the larger number of electron rich atoms (i.e. non-hydrogen atoms for simplicity). eg

While both electrophilic additions are possible, the more favoured product is always the one that has the more stable carbocation and so the bromide ion reacts there (Markovnikov rule).
© Copyright rmcstudy
