Reactions and Theory of Acids
Acids are a source of hydrogen (H+) ions.
Arrhenius definition of acid : A substance that dissociates in water to give hydrogen ions (H+).
Arrhenius definition of base : A substance that when dissolved in water dissociates to produce hydroxide ions (-OH).
Water forms a dative covalent bond with the H+ ion to form the hydronium ion (H3O+), which technically is what an acid is (below, A just represents a generic part of a molecule which forms a negative ion after dissociation such as Cl or NO3 for example).

Bronsted-lowry theory: acids are proton donors, bases are proton acceptors.
Conjugate pairs form because when an acid loses a proton, it becomes a base in the reverse reaction and when a base gains a proton, it becomes an acid in the reverse reaction. An acid-base reaction is an equilibrium between two acid-base conjugate pairs.

Common reactions of acids:

pH
H+ concentration ([H+]) in solutions can range from ~10 mol dm-3 to ~10-15 mol dm-3.
pH scale is a more convenient way of measuring [H+].
Table of pH vs [H+ (aq)]

As the concentration of H+ ions will usually involve very inconvenient numbers, a logarithmic scale is used so we can have a more convenient method of measuring acidity (H+ concentration).
pH = -log[H+ (aq)]
The ionic product of water (Kw) arises from the fact that water dissociates (to a very small extent) according to the equation: H2O <=> H+ + OH-. As this reaction is in equilibrium (with the position of equilibrium lying very much to the left) the equilibrium constant, Kc can be calculated. The concentration of water is constant (see video) and will barely change above, so we can rearrange the Kc equation (Kc = [H+ (aq)][OH-(aq)] / [H20]) to become Kc x [H2O] = [H+ (aq)][OH-(aq)] and as the left hand side is a constant, we can call it a new name: Kw, the ionic product of water). This can be measured and is found to be 1.00 x 10^(-14) at room temperature (it will vary with temperature as all Kc values vary with temperature).
Kw controls the balance between the [H+ (aq)] and the [OH-(aq)] in all aqueous solutions; relative concentrations of each are determined by Kw.
For pure water, [H+] = [OH-], so we can calculate [H+], as [H+]x[H+] = 1.00 x 10^(-14). so taking the square root of both sides [H+] = 1.00 x 10^(-7) and so pH = 7.
To calculate the pH of a weak acid, the acid dissociation constant above has to be used.

and pH = -log[H+]
Indicators & Titration curves
Many indicators are weak acids ( partially dissociate in aqueous solution).
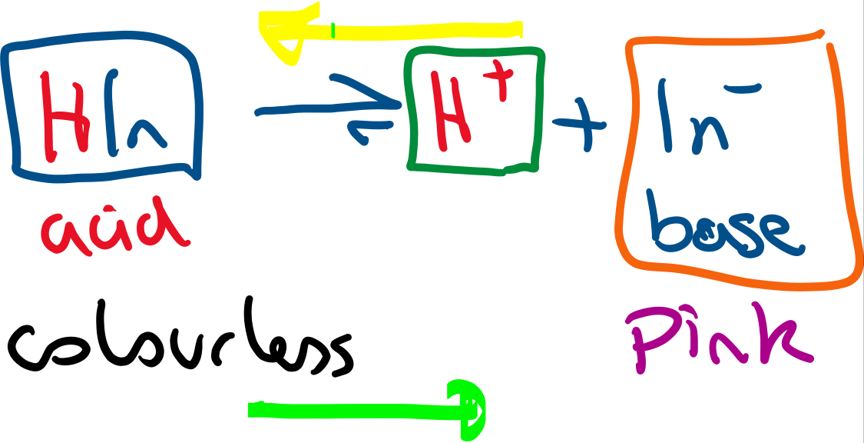
Above is an equilibrium depiction for phenolphthalein. As this is an equilibrium, if acid is added to the indicator solution, the position of equilibrium shifts to the left (yellow arrow) and the solution becomes colourless. As alkali is added, the H+ concentration decreases and so the position of equilibrium shifts to the right (green arrow) and the solution becomes pink (pale purple).
All indicators have a different Ka values, they will change colour at a different pH.
Titration curves are graphs created when an alkali is added slowly to the acid and the reaction is monitored using a pH meter.
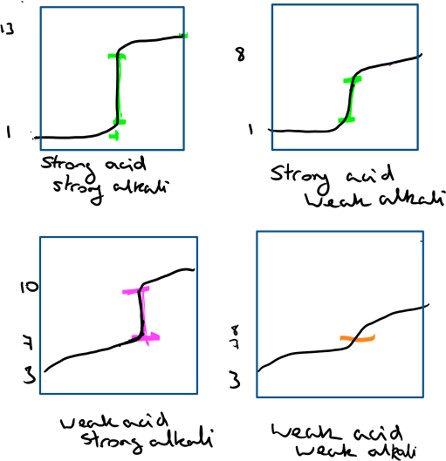
In each of the reactions above, the completion point occurs at a different pH, due to the amount of alkali (hydroxide ions) added at the point of reacting with all of the hydrogen ions. An indicator will only work if the pH at which it changes colour (its range) coincides with the equivalence point in each reaction (mid point of the above vertical ranges).
© Copyright rmcstudy