Introduction to equilibrium
A few definitions are necessary to begin with:
Reversible reaction – reaction where products can under certain conditions turn back into reactants.
Closed system – chemical system where no physical substances can be added or removed, energy can be transferred in and out.
Dynamic equilibrium – occurs when a chemical system has reached a state where there are constant amounts of reactants and products, & the rate of forward reaction is equal to the rate of the backward reaction.
Position of equilibrium (POE)– gives a reference to the various proportions of reactants or products in the chemical system. right = more products, left = more reactants.
Le Chatelier's Principle
Le Chatelier’s principle – if a dynamic equilibrium is disturbed by a stress, such as changing conditions of temperature, pressure or concentration, then the reaction moves to oppose the stress.
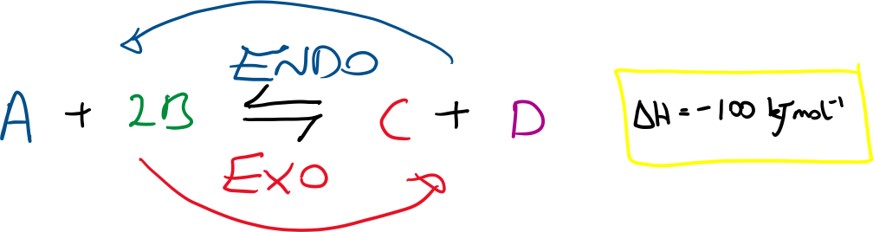
Concentration:
If [A] increases then the system will shift to counteract the change and so will reduce the amount of A by reacting it with B to form more C + D thus shifting the position of equilibrium to the right (i.e. more products are formed as the forward reaction is favoured).
Pressure:
If the pressure is increased then the system will shift to counteract the change and so will attempt to decrease the pressure by favouring the side of the reaction that produces the fewest number of moles, i.e. producing more of C + D, 3 moles > 2 moles. The position of equilibrium shifts to the right (i.e. more products are formed as the forward reaction is favoured).
Temperature:
If the temperature is increased then the system will shift to counteract the change and so will attempt to decrease the temperature by favouring the side of the reaction that produces less heat, i.e. the endothermic reaction and so producing more of A + 2B. The position of equilibrium shifts to the left (i.e. more reactants are formed as the backward reaction is favoured).
Industry
Haber Process:
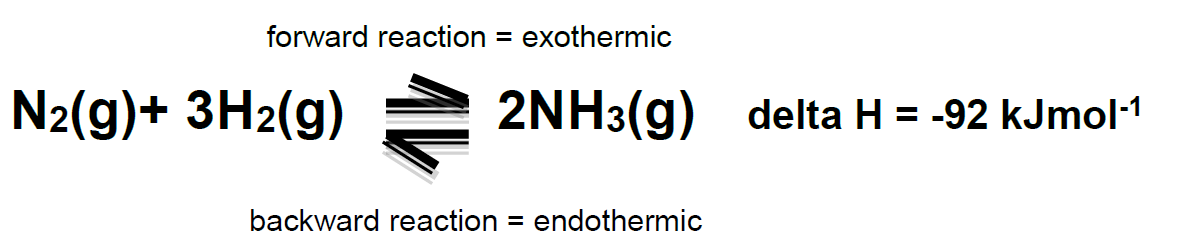
Conditions which affect the haber process and compromises made to increase production of ammonia:
Decrease in temperature - System moves to increase temperature by favouring the exothermic reaction as this releases heat energy. POE moves to the right, increasing the amount of ammonia produced.
Increase in pressure - System moves to decrease pressure by favouring the reaction that produces fewest gas molecules. POE moves to the right, increasing the amount of ammonia produced.
The actual conditions used to in the Haber process are a compromise. Ideally a low temperature and a high pressure (as discussed above) would favour a high yield of ammonia. However, the reaction would be too slow with a low temperature so ~500 C is used. A high pressure would be dangerous and expensive, so 200 atm is used. An iron catalyst is used to increase the rate at which equilibrium is formed.
Contact process:
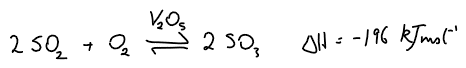
Sulfur dioxide reacts with oxygen (equilibrium process above) with the aid of a Vanadium (V) Oxide catalyst to form sulfur trioxide.
Sulfur trioxide then reacts with sulfuric acid to form a compound called oleum which when added to water forms sulfuric acid. (If sulfur trioxide is added directly to water an extremely corrosive fog of sulfuric acid is formed, so dissolving it in pre-made concentrated sulfuric acid is most manageable.
For the above reaction, high pressure and low temperature would favour the equilibrium production of sulfur trioxide but the high pressure is uneconomical and the low temperature is too slow. A compromise of atmospheric pressure and a temperature of 450 C gives a very high yield of sulfur trioxide.
Equilibrium Law ~ Kc
For any general reaction shown below:

The equilibrium constant (Kc) is as follows:
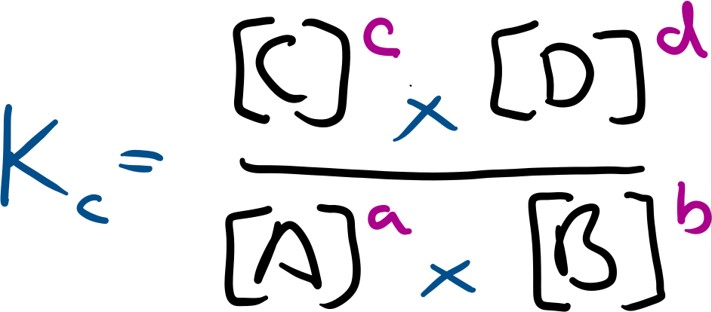
Kc is a constant at constant temperature ( for more on how Kc changes with temperature, you will need to look into Gibbs free energy, which I have not covered on this site thus far). Kc controls the amounts of reactants and products in the equilibrium. If the concentration of something is changed then the proportions of reactants and products change so as to keep the Kc constant. If more reactants are added, then the amount of products must increase so as to maintain a constant Kc.
© Copyright rmcstudy