rate of reaction = change in concentration of reactant or product divided by the time it takes for the change to take place.![]()
As reactions proceed fewer collisions take place per second and so the reaction slows down.
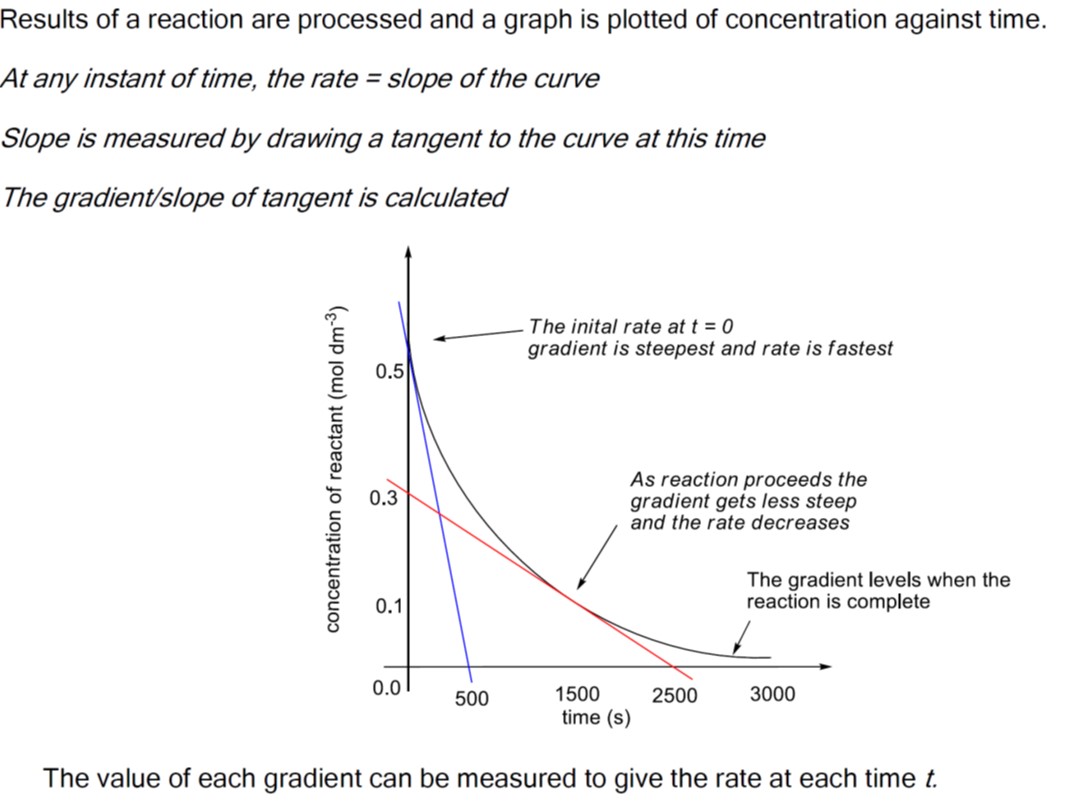
Remember that the rate of change can be calculated using the formula for the slope of a line in coordinate geometry, i.e. the difference in the y-values divided by the difference in the x-values.
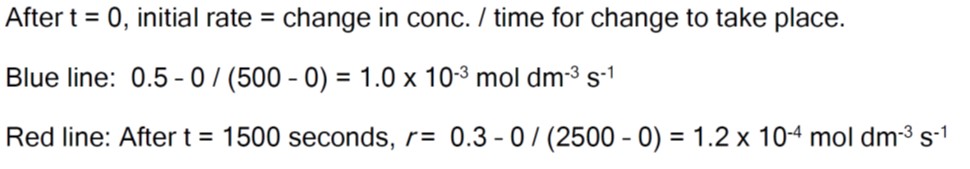
*hence rate is higher (reaction faster) at the beginning.
© Copyright rmcstudy