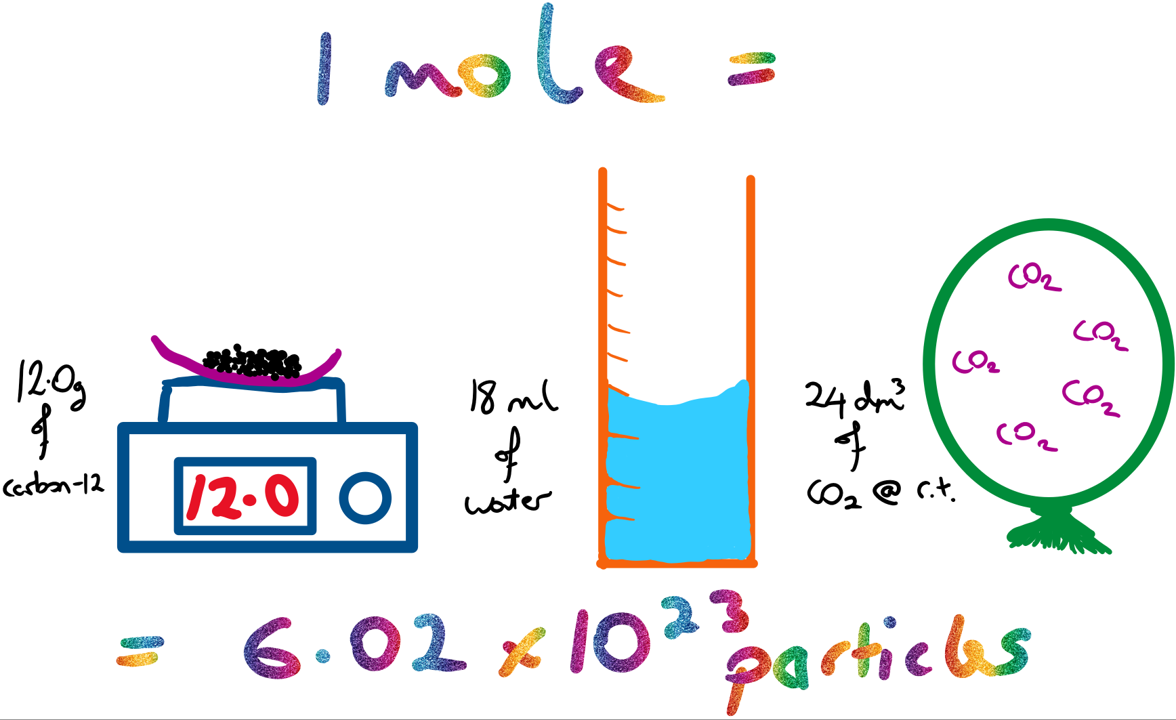
A mole is a unit that refers to a specific amount of a chemical substance. One mole (1 mol) is the amount of a substance that contain![]() particles (either atoms, molecules or formulae) of the substance. e.g. in 1 mol of water there are
particles (either atoms, molecules or formulae) of the substance. e.g. in 1 mol of water there are![]() water molecules, in 1 mol of helium there are
water molecules, in 1 mol of helium there are ![]() atoms of helium (= Avogadro’s number).
atoms of helium (= Avogadro’s number).
RAM - Relative Atomic Mass (Ar) = the average mass of an atom compared to 1/12th the mass of an atom of carbon-12.
e.g. Chlorine mainly contains 2 isotopes: Cl-35 (75%) & Cl-37 (25%)
The Ar is an average of the two taking into account relative abundances:(35 x 0.) + 37 x 0.25) = 35.5
RFM - Relative Formula Mass (Mr) = sum of RAMs of all atoms present in a formula
e.g. Mr of H2SO4: (1 x 2) + (32 x 1) + (16 x 4) = 98
Ar & Mr are the symbols of RAM and RFM respectfully.
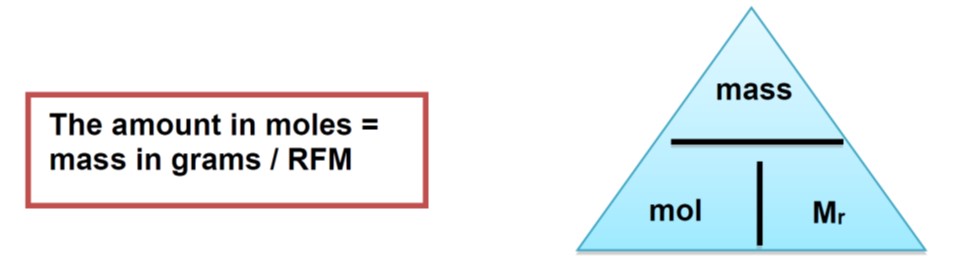
The mass of one mole of atoms/molecules is the RAM/RFM expressed in grams.
To work out the empirical formula of a compound, you need to compare the number of moles of each element present in the compound. Therefore, to do this you first take the percentage or (mass in g) of each element present to work out the number of moles of each. Divide the number of moles of each element by the smallest value (number of moles) as this will will result in the simplest whole number ratio.
e.g.:


Not as clearly identified in the video is the method to quickly determine the volume of a gas when the number of moles is known. At r.t.p (room temperature & pressure, ~25C and 1 atm): volume (dm3) = mol x 24, and at s.t.p (standard temperature and pressure, = 0C and 1 atm): volume (dm3) = mol x 22.4
© Copyright rmcstudy