Hess’ law: If a reaction can take place by more than 1 route and the initial and final conditions are the same, the total enthalpy change is the same for each route.
A Hess cycle is a diagram showing alternative routes between reactants and products which allows the indirect determination of an enthalpy change from other known enthalpy changes using Hess’ law.
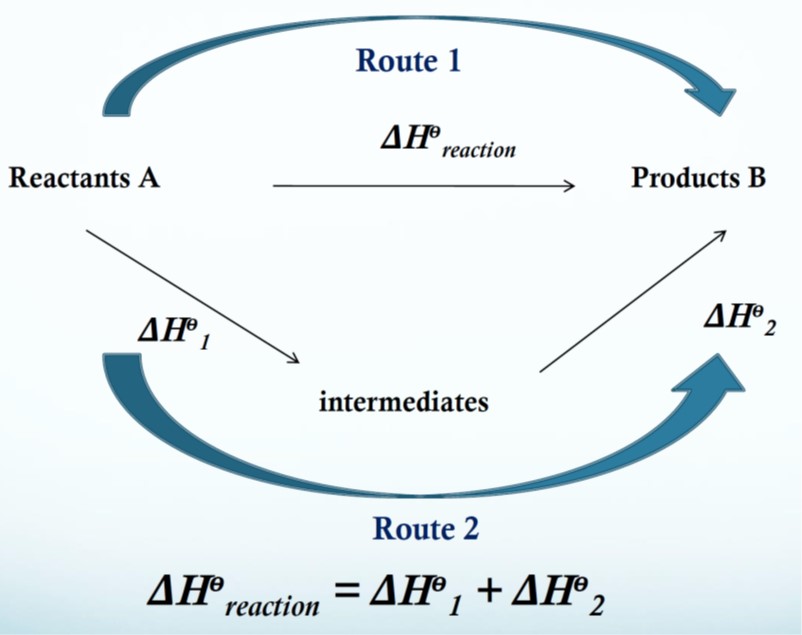
If you know two of the energy changes for the above reactions then you can calculate the third.
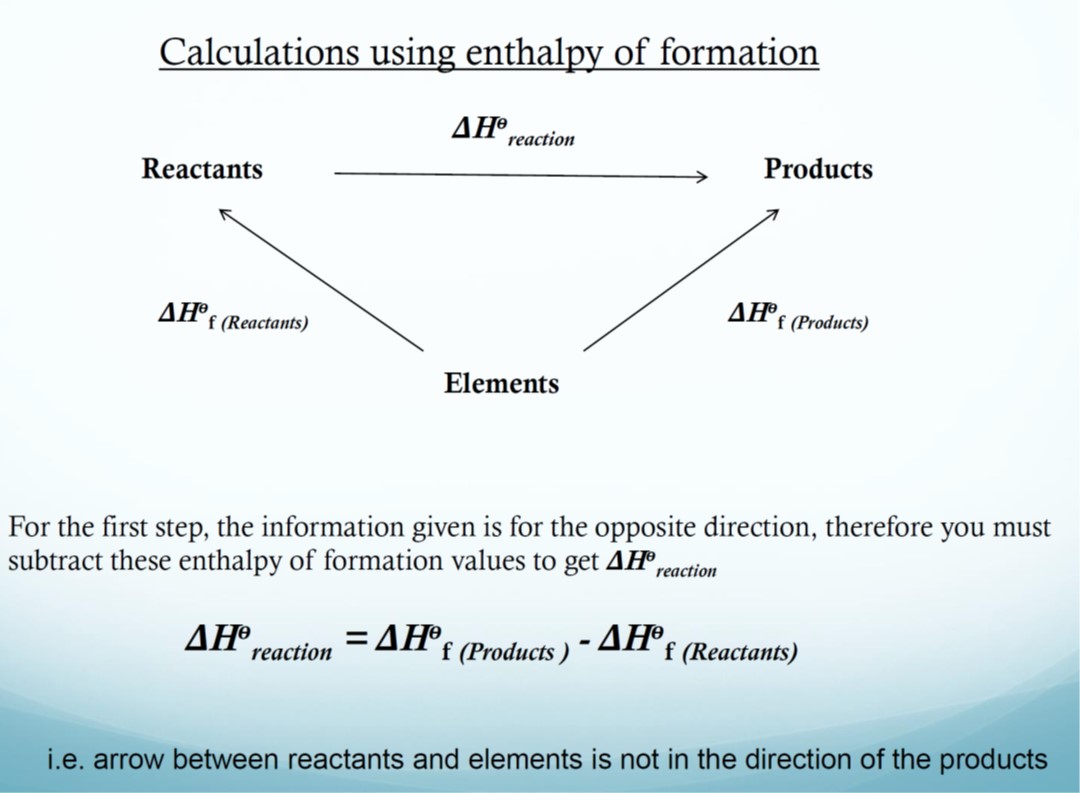
*Heat of formation, ΔHf
The heat of formation is the energy change that takes place when 1 mole of a compound is formed from its constituent elements in their standard states under standard conditions.
The heat of formation of an element in its standard state is zero.
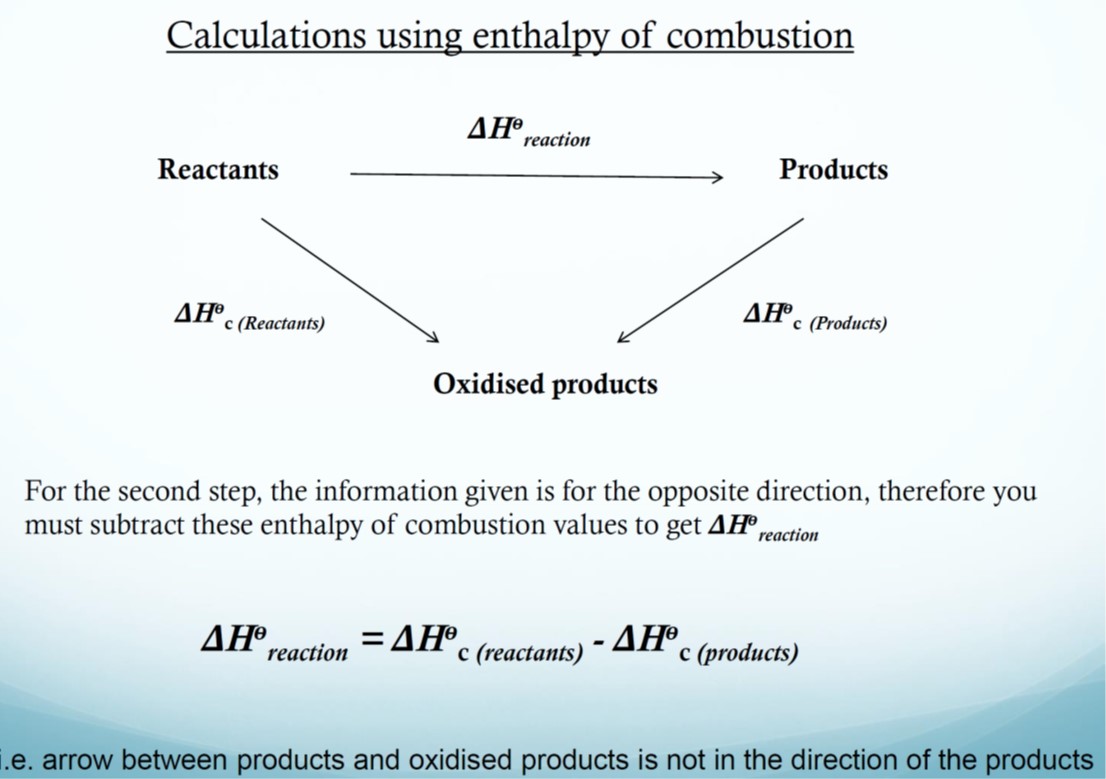
Heat of combustion, ΔHc
The heat of combustion is the energy change that takes place when 1 mole of a substance reacts completely with oxygen under standard conditions, all reactants and products being in their standard states.
© Copyright rmcstudy