Many indicators are weak acids (– partially dissociate in aqueous solution).
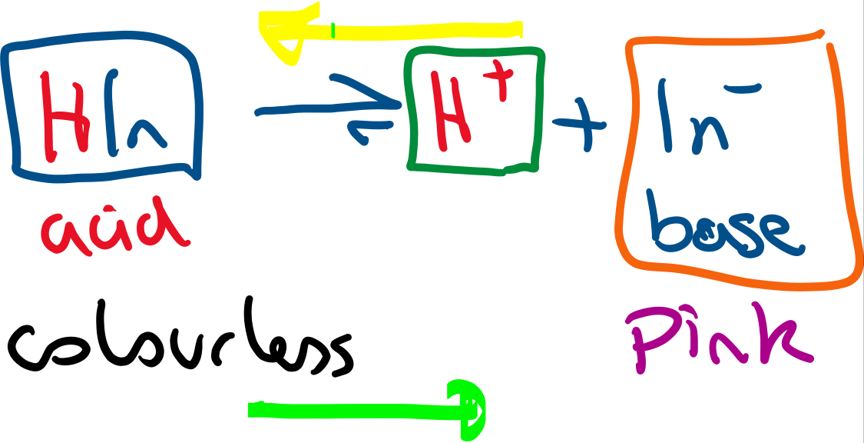
Above is an equilibrium depiction for phenolphthalein. As this is an equilibrium, if acid is added to the indicator solution, the position of equilibrium shifts to the left (yellow arrow) and the solution becomes colourless. As alkali is added, the H+ concentration decreases and so the position of equilibrium shifts to the right and the solution becomes pink (pale purple).
All indicators have a different Ka values so they will change colour at a different pH.
Titration curves are graphs created when an alkali is added slowly to the acid and the reaction is monitored using a pH meter.
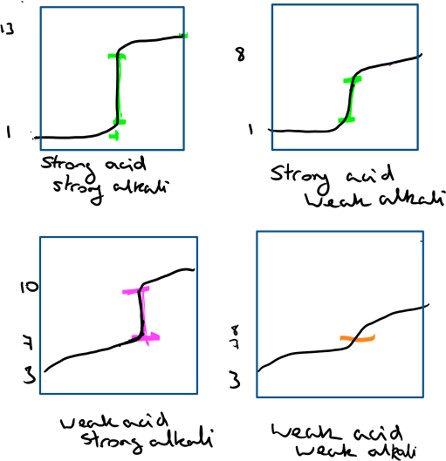
In each of the reactions above, the completion point occurs at a different pH, due to the amount of alkali (hydroxide ions) added at the point of reacting with all of the hydrogen ions. An indicator will only work if the pH at which it changes colour (its range) coincides with the equivalence point in each reaction (mid point of the above vertical ranges).
© Copyright rmcstudy