H+ concentration ([H+]) in solutions can range from ~10 mol dm-3 to ~10-15 mol dm-3.
pH scale is a more convenient way of measuring [H+].
Table of pH vs [H+ (aq)]

As the concentration of H+ ions will usually involve very inconvenient numbers, a logarithmic scale is used so we can have a more convenient method of measuring acidity (H+ concentration).
pH = -log[H+ (aq)]
The ionic product of water (Kw) arises from the fact that water dissociates (to a very small extent) according to the equation: H2O <=> H+ + OH-. As this reaction is in equilibrium (with the position of equilibrium lying very much to the left) the equilibrium constant, Kc can be calculated. The concentration of water is constant (see video) and will barely change, so we can rearrange the Kc equation (Kc = [H+ (aq)][OH-(aq)] / [H20]) to become Kc x [H2O] = [H+ (aq)][OH-(aq)] and as the left hand side is a constant we can call it a new name: Kw, the ionic product of water). This can be measured and is found to be 1.00 x 10-14 at room temperature (It will vary with temperature as all Kc values vary with temperature).
Kw controls the balance between the [H+ (aq)] and the [OH-(aq)] in all aqueous solutions; relative concentrations of each are determined by Kw.
For pure water, [H+] = [OH-], so we can calculate [H+], as [H+]x[H+] = 1.00 x 10-14 . so taking the square root of both sides [H+] = 1.00 x 10-7 and so pH = 7.
To calculate the pH of a weak acid, the acid dissociation constant above has to be used.
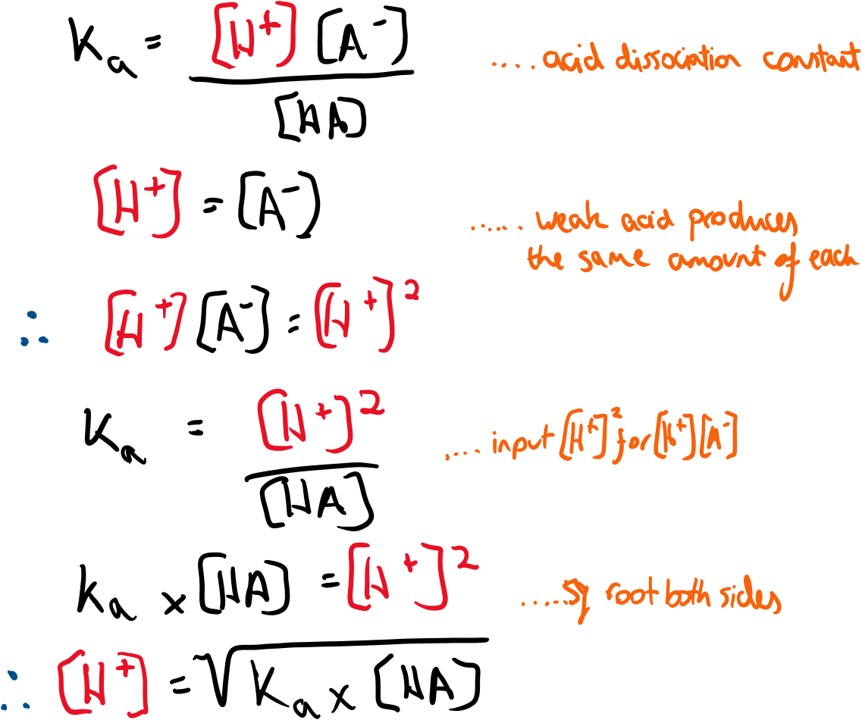
and pH = -log[H+]
© Copyright rmcstudy